Linguistic Validation Explained: the Process, Regulation and What Your Trial Actually Needs
Apr 11, 2026
5
min read

Clinical outcome assessments (COAs) are only as reliable as the language used to collect them. When patients across dozens of countries answer the same questionnaire in different languages, the integrity of your endpoint depends entirely on whether those language versions are measuring the same underlying construct. Linguistic validation (LV) is the structured, evidence-based methodology that makes that comparability possible – and protecting it is the core of what we do at Borliner.
The problem LV exists to solve
Multinational clinical trials routinely deploy COAs – PROs, ClinROs, ObsROs, PerfOs – across 20, 40 or more languages. Each of those language versions is a potential source of measurement error if it has not been properly adapted and validated. The risk is not poor grammar or awkward phrasing – linguistic validation goes far beyond linguistic perfectionism. The risk is construct drift: patients in different countries interpreting the same item differently, making the pooled data less reliable or even incomparable.
Consider a fatigue item used across German, Arabic and Spanish-speaking populations. "Fatigue" does not map cleanly to a single concept across those cultures and languages. Without linguistic validation, each language team resolves the ambiguity independently – and the endpoint begins to splinter.
This is not a theoretical concern. It is a well-documented source of variability in multinational data, addressed directly by ISPOR, ISOQOL and FDA guidance. Linguistic validation is the industry's answer to it.
What linguistic validation is – and what it is not
Linguistic validation is not translation – well, not merely translation. Standard translation workflows (typically translation, editing and proofreading (TEP)) are optimized for fluency, terminology and stylistic accuracy. These are necessary but insufficient for COA instruments.
LV adds a layer of scientific rigor that TEP cannot provide:
- Conceptual equivalence review – ensuring that the construct being measured survives the transition into the target language and culture
- Cultural appropriateness assessment – identifying idioms, lifestyle references, time frames or social norms that do not map onto the target population's lived reality
- Patient-level comprehension testing – directly verifying, through structured cognitive interviews, that patients understand each item as intended
ISPOR's foundational good-practice guidance frames LV as a process of achieving four types of equivalence simultaneously: semantic, conceptual, idiomatic and experiential. Missing any one of them introduces systematic bias – not random noise, but directional error that can distort endpoint results in ways that are difficult to detect or correct after the fact.
The Regulatory Landscape
Linguistic validation is not best practice only but increasingly a regulatory expectation.
The core reference documents shaping current requirements are:
- ISPOR "Principles of Good Practice" for translation and cultural adaptation of PROs – the foundational methodology standard, dating back to 2005, recommending dual forward translation, reconciliation, back-translation, expert review and patient cognitive debriefing
- ISPOR Translation & Linguistic Validation Task Force report – covering language selection decisions and methods for supporting data pooling when multiple language versions are used
- ISOQOL "Principles of good practice for translation of eCOA" – integrating translation, cultural adaptation and electronic implementation as a single, concurrent process
- FDA PRO Guidance and PFDD Guidance 3 – making clear that evidence for label claims must include demonstration that all language versions of an instrument are fit for purpose for their respective populations
- ISOQOL "Good practices for LV of ClinRO, ObsRO, and PerfO measures" – extending standardized LV methodology to the non-PRO COA types that previously lacked formal guidance
Collectively, these documents establish that a sponsor seeking a PRO-supported label claim in a multinational submission must be able to demonstrate that all language versions of the instrument are content-valid, culturally appropriate and adequately documented. The linguistic validation report is the evidentiary artifact that supports that demonstration.
The LV process: what each step accomplishes
The LV workflow has remained largely consistent since ISPOR's foundational guidance, and for good reason: each step addresses a specific failure mode.
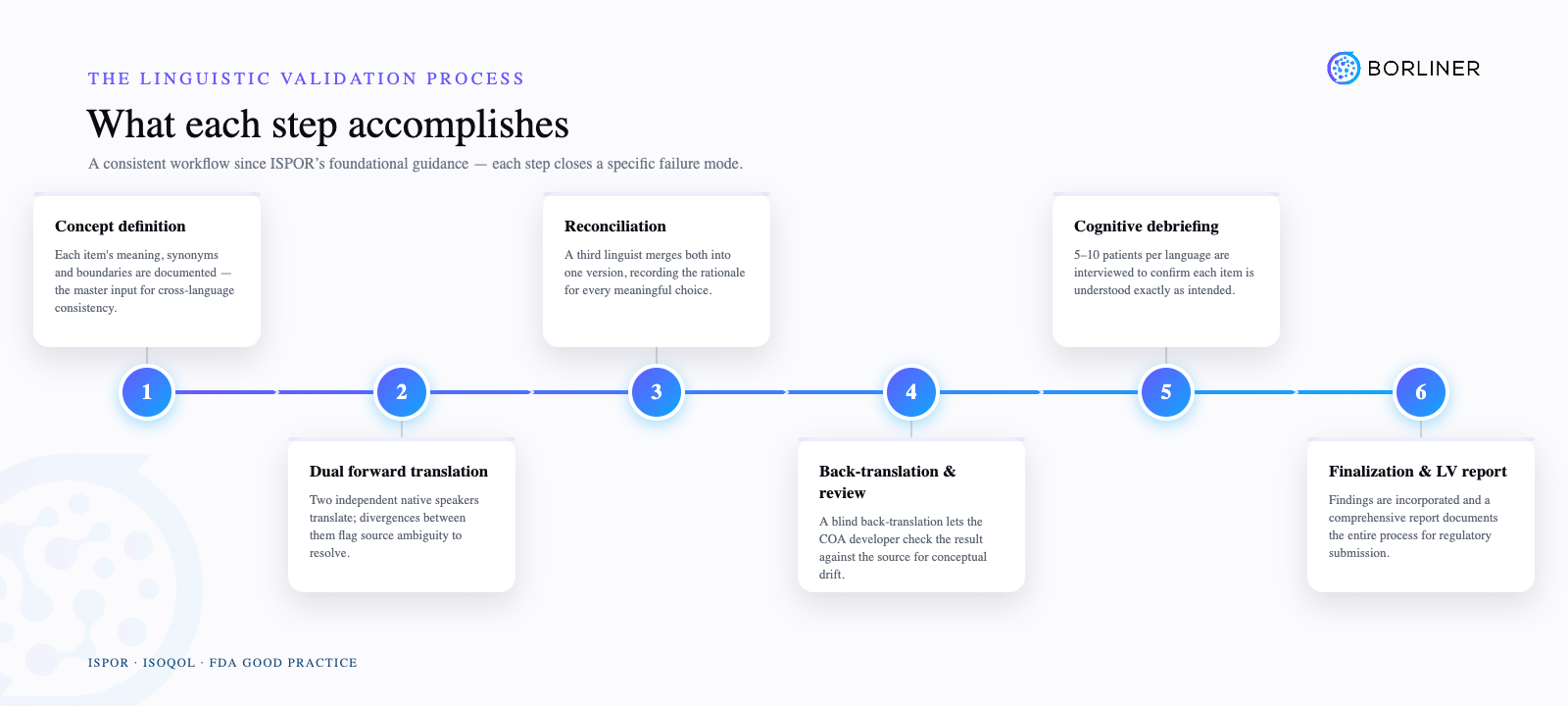
Concept definition
Before translation begins, each item is documented with its intended meaning, acceptable synonyms, construct boundaries and any terms that should be avoided. This document is the single most important input to cross-language consistency. Without it, translators in each language independently resolve ambiguities – and produce translations that are internally coherent but cross-linguistically inconsistent.
Dual forward translation
Two independent native-speaker translators produce separate translations, whereby the divergences between them are not problems but diagnostic signals. Every point of divergence is a place where the source instrument's meaning was ambiguous enough to generate different valid interpretations. That ambiguity must be resolved before it enters a patient's hands.
Reconciliation
A third linguist reviews both forward translations and produces a single unified version. Critically, this linguist is different from both forward translators, eliminating the bias toward one's own prior decisions. The reconciliation document records the rationale for every meaningful choice – this is regulatory-facing documentation, not an internal working file.
Back-translation and developer review
An independent translator (or two translators) renders the reconciled version back into the source language, blind to the original text. The COA developer or copyright holder compares the result against the source instrument. A clean back-translation does not certify quality, but a problematic one flags conceptual drift that might otherwise go undetected by non-native reviewers.
Cognitive debriefing
One-on-one structured interviews with 5–10 patients per language, probing comprehension of each item: what does this question mean to you? What do you think it is asking? The interviewer does not lead or correct – the goal is to capture how a representative patient actually processes each item, not how they should process it.
This is where validation happens in the truest sense. Expert review can identify many problems. Only patient testing can confirm – validate! – that the instrument works as intended for the people who will actually be completing it.
Finalization and LV report
Interview findings are incorporated, the final version is approved, and a comprehensive report documents the entire process – translations, back-translations, cognitive debriefing summaries, decision rationales and any outstanding issues with resolutions. This report is what regulators and HTA bodies expect to see.
Linguistic validation and eCOA: a new set of requirements
The shift from paper to electronic COA administration has introduced a category of measurement risk that paper-era LV guidance was not designed to address. Screen size, text truncation, font rendering, navigation flow and device differences can all alter how a patient reads and responds to an item – even when the underlying translation is accurate and validated.
ISOQOL's eCOA good-practice guidance establishes a clear principle: translation, cultural adaptation and electronic implementation must be treated as concurrent processes, not sequential hand-offs. A translated and validated paper instrument is not automatically validated in its eCOA form.
Best practice for eCOA linguistic validation now includes:
- Electronic Language Feasibility Assessment (ELFA) – evaluating how translated text will render on target devices before design is finalized, identifying truncation risks, text expansion issues and layout conflicts
- Screenshot proofreading – screen-level quality control of all language versions in the actual eCOA environment, not just document-level review
- Concurrent planning with COA copyright holders and eCOA vendors from the point of instrument selection, not at study start-up
At Borliner, eCOA migration support is a core part of our LV service offering, precisely because the migration step is where well-validated paper instruments most commonly lose their measurement integrity.
Beyond PROs: LV for ClinRO, ObsRO and PerfO
For years, formal LV methodology guidance focused almost exclusively on PRO instruments. Clinician-reported (ClinRO), observer-reported (ObsRO), and performance (PerfO) outcome measures operated largely without equivalent standardization.
ISOQOL's 2020 good-practice document on LV of non-PRO COA types addressed this gap directly. The core translation process – dual forward translation, reconciliation, back-translation, expert review – largely mirrors PRO methodology. The key distinction lies in the cognitive debriefing step: for ClinRO instruments, the relevant respondents are clinicians, not patients, and the interview approach must be adapted accordingly to probe clinical interpretation and professional usage rather than patient comprehension. Clinician cognitive debriefing, however, has been noted as “as an acceptable but not mandatory approach, which could be supplemented or replaced by a clinician review of the translation in most cases.”
Operational Risks of Inadequate LV
For sponsors and CROs evaluating LV as a budget line item, it is worth mapping the specific failure modes against the associated costs:
- Non-comparable data across countries – Different patient interpretations inflate variance, reduce statistical power and may require post-hoc explanations that weaken regulatory submissions
- Regulatory questions at filing – Missing or inadequate LV documentation for PRO-based endpoints can trigger requests for additional evidence at the point of highest submission pressure
- Late-stage wording amendments – Errors discovered near database lock require urgent site communications, data management interventions and potentially protocol amendments – costs that dwarf the original LV investment
- Undermined HTA arguments – HTA bodies and payers scrutinize PRO-based claims; inadequate translation documentation can erode confidence in the entire dataset and affect reimbursement outcomes
Well-executed LV is a relatively small upfront investment against each of these risks. For programs with PRO-based primary endpoints running across 40 or more languages, the cost of LV is a fraction of a percent of total trial spend – and its absence can threaten the submission itself.
Frequently Asked Questions
Can we reuse translations from a previous study?
Previously validated translations can and should be reused across studies. The prerequisite is that the original LV documentation exists and is traceable – because regulators may request evidence of the validation process. Where no documentation exists, there is no way to demonstrate the translation was properly validated and a gap assessment would be needed before relying on it in a submission.
Does back-translation guarantee quality?
No. ISPOR guidance explicitly states that back-translation alone cannot ensure conceptual equivalence or confirm patient comprehension. It is a useful diagnostic tool, but definitely not a substitute for the full LV process.
Do we need LV for non-English source instruments?
Yes. The same methodology applies regardless of the source language. Conceptual equivalence and cultural appropriateness must be established in every language cohort, including when translating from French, German, Japanese, or any other source. Still, there are a couple of approaches to validating the non-English source instruments with no final guideline published up to date.
How early in the trial should LV be planned?
As early as COA selection and protocol design. By the time a protocol is locked, many LV-related decisions – including instrument selection, language scope and eCOA platform – have already been made. Involving LV expertise upstream reduces rework and prevents downstream measurement problems.
How Borliner supports the full LV lifecycle
Borliner is a specialized provider of linguistic validation services, working with CROs, copyright holders and global translation companies across more than 200 languages. Our services span the complete LV lifecycle:
- Cognitive debriefing – patient and clinician cognitive interviews with structured analysis and full debriefing reports
- Patient and clinician recruitment – targeted recruitment of respondents matching the instrument- and developer-specified population for cognitive debriefing
- Clinician reviews – structured reviews for ClinRO, but also for PRO and ObsRO instruments
- eCOA migration support – screenshot proofreading and screen-level QC
- Full-lifecycle LV project management – coordinating translation vendors, copyright holders and eCOA platforms within a single, documented workflow
Our work is grounded in ISPOR, ISOQOL and FDA good-practice standards and our documentation is designed to support regulatory submissions from the outset.
Want to learn more or discuss? Get in touch.
References
ISPOR Translation & Linguistic Validation Task Force report for multinational trials
ISOQOL Principles of Good Practice for Translation of Electronic COAs
Considerations for Updating Existing Linguistically Validated Clinical Outcome Assessments (COAs)
Contact Us
Speed your research up,
contact us today
Email us to get a quote in record time or book a call if you have any other questions or custom requests.


